My last blog post “Don’t Overlook MBD over the Web” concluded the second aspect of Table 1 on Process. From our first MBD Implementation blog post last October to today, this series of Model-Based Definition (MBD) Implementation 10 DOs and 10 DONTs has run over the last five months. I’m glad that I didn’t give it up despite so many competing priorities, even if it means writing on weekends.
Thank you for your interests, support, comments, and sharing in the past five months. You are the reason behind this series. MBD topics have gained strong attraction in recent years: each blog post in this series has organically reached tens of thousands of viewers on average.
Now let’s carry onto the last aspect: Product, focusing more on specific product design and manufacturing issues. This is the meat for us designers and engineers, or hands-on doers: exciting! First suggestion is to pilot with one manufacturing document. Again your comments below are always welcomed.

In a product lifecycle, there is an ocean of manufacturing documents. In an initial MBD implementation, don’t try to boil that ocean or test MBD everywhere. Instead, let’s focus on one case to ignite a spark that can shed inspiring light and set up the foundation for future expansion. Breakthrough always happens at the weakest spot of an obstacle, so our job is to identify this spot.
Then how do you pick a favorable breakthrough point in an MBD implementation? In general, I think we want an angle where:
- All key stakeholders agree to experiment to avoid any sabotage
- 3D is already taken as the design authority over 2D
- 2D drawings have become less important
- 2D drawings have even become a frustration
- Potential benefits are remarkable (high-reward upside)
- Potential failure is affordable and manageable (low-risk downside)
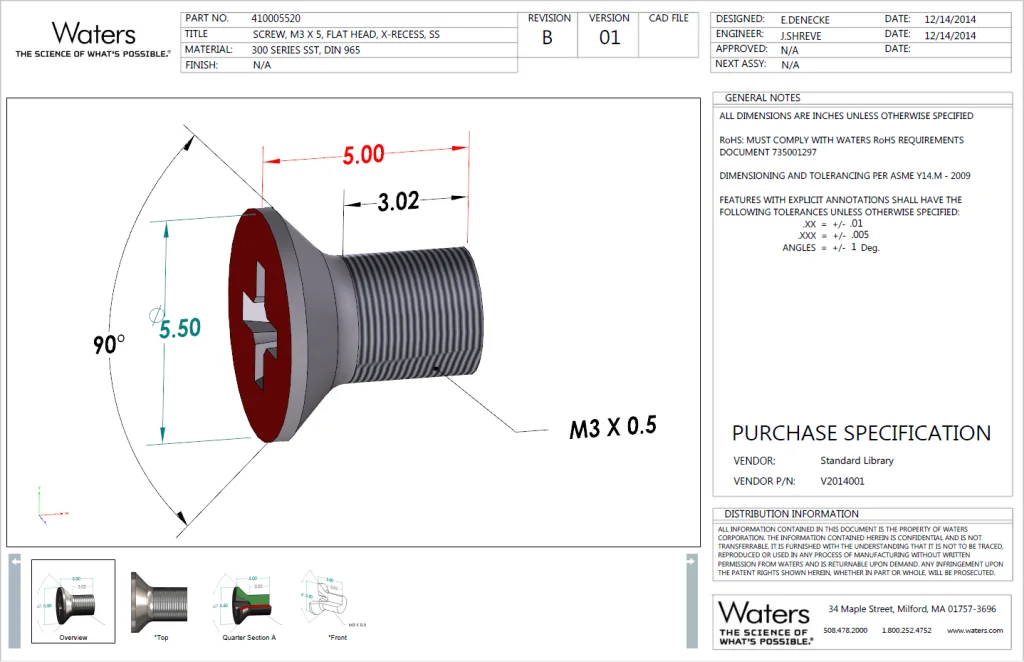
For example, Waters Corporation started with purchased and/or standard part (nuts, bolts, and pipes) specifications, which have only minimal or critical dimensions as shown in the image below.
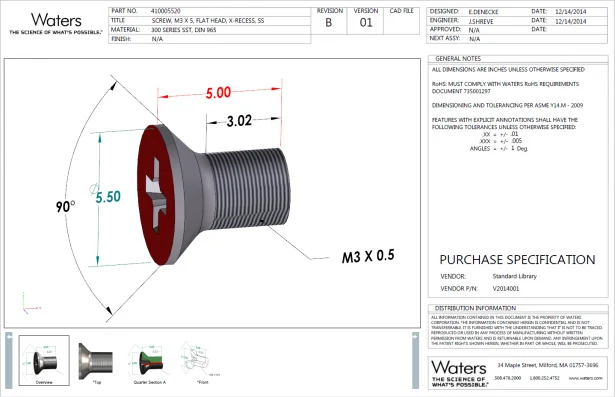
Internally, there has been a long and on-going debate as to why to create full drawings for them. Purchasing and Incoming Inspection teams always insisted on a drawing, or they would have nothing to validate parts against. But in reality, these components are manufactured per industry standards anyway, so most of the critical information that Purchasing and Incoming Inspection need can be presented in title blocks such as Part Number, Title, Material, and Finish. Besides, general tolerances can be specified in Notes, such as .xx = +/-.01, xxx = +/- .005, ANGLES = +/- 1 Deg. Therefore, SOLIDWORKS MBD 3D PDF provided a perfect solution. It’s a win-win: this new process can reduce 2D drawings and other teams have a richer and more intuitive document to validate. Most importantly, it raises the visibility of MBD and 3D PDFs to set up next step.
Along the same lines, a consumer product manufacturer picked organically shaped plastic part specifications as an MBD pilot, because these organic curves and shapes are very cumbersome to individually define in 2D drawings anyway. Instead they just define 3D profile tolerances for injection molding and then use CMM or 3D scanning for inspection.
Another medical device company found 3D PDF serves Request For Quote (RFQ) well. Suppliers can understand them much more easily with better accuracy. Even a sales guy on the road can view models, dimensions, tolerances using free Adobe Reader, and probably provide a quote right away. Now the quoting time is much shorter.
That’s it for now in this blog. Next one will continue this Product aspect discussion: Don’t skip calling out critical to function 3D PMI. Missing critical to function dimensions and tolerances has left MBD a bad reputation in some suppliers’ experiences. We will discuss the reasons behind these negative experiences and how to avoid unnecessary resistance to MBD. To learn more about SOLIDWORKS MBD, please visit its product page. Also welcome to discuss with me at Twitter (@OboeWu) or LinkedIn (OboeWu).