Manufacturers who are growing faster than competitors and gaining market share while thwarting price wars all share a common trait. They have an intensity about product quality that reverberates through each work center, plant, and across their production network.
Flat screen monitors in work areas show the latest quality metrics they’re tracking, SPC charts are on additional flat screens and message boards. Intensity about quality and excelling for customers dominates their cultures and focus.
- Managing to quality metrics provides a goal-driven roadmap for excelling and exceeding compliance standards which is a strong competitive advantage.
- Quality metrics are interdependent on and reflect the relative levels of performance of other functional areas of the plant, and are also leading indicators of production performance.
- Attaining compliance with challenging industry-specific standards including TS (automotive), 21 CFR Part 11 (medical products and aerospace & defense) and ISO standards while reducing the cost of quality is achievable when quality and compliance goals are customized to each plant.
Great Teams Know That Excelling At Quality Earns Customers For Life
Creating and sustaining a passion for quality brings greater responsiveness and flexibility into manufacturing operations company-wide. By concentrating on a core set of quality metrics, manufacturing teams have the goals they need to focus on.
The greatest achievements in quality are happening today because every team member, operator and worker has ownership of outcomes.
Across the spectrum of manufacturing strategies, from highly customized Engineer-to-Order (ETO) to Make-to-Order (MTO) and Make-to-Stock (MTS) quality improves when every member of a team sees how their efforts make a difference.
Quality Is The Strongest Force Multiplier For Growth A Company Has
Attaining higher quality levels has a multiplicative effect throughout the entire company. Finding new ways to improve perfect order performance continually reduces Return Material Authorizations (RMA) and increases customer satisfaction leading to more sales.
Improving Overall Equipment Effectiveness (OEE) reduces manufacturing cycle times and also contributes to improving perfect order performance, meeting customer commitments on time. Managing OEE to the highest levels possible has a direct impact on production yields while also reducing the cost of quality, further driving down costs per unit of production excluding materials.
All of these factors taken together improve gross contribution margins while meeting or exceeding customer expectations as defined by committed to delivery dates. When all of these factors are taken together in the context of compliance to 21 CFR Part 11 in medical products for example the competitive strengths are clear.
Successful manufacturers who grow despite industry conditions have found a way to turn compliance into a competitive advantage. Unlike their competitors, compliance is seen as more than a cost. It’s perceived as a means to differentiate and excel at every level of manufacturing. The world’s most successful manufacturing companies perceive compliance standards as liberating goals to be met and exceeded.
5 Key Metrics That Drive Higher Manufacturing Quality
Each of the five key metrics reflects how interdependent quality is across manufacturing operations. When they are combined with additional financial and operations data, each provides added insight into how quality can improve.
A key takeaway of these five metrics taken in total is that quality must be a unifying thread across all manufacturing operations to succeed:
1. Corrective Action/Preventative Action (CAPA) events
Also known as Corrective Action Requests (CAR) addressing this metric and the events they define are required to stay in compliance with rigorous quality standards that vary by industry.
An example is FDA 21 CFR 820.100 for medical device manufacturers that require their quality management systems have CAPA tracking, reporting and auditing accessible on a 24/7 basis by FDA auditors.
2. Engineering Change Order (ECO) Tracking
Also known of as Engineering Change Notice (ECN) this metric tracks changes to routing, Bills of Materials (BOM), production workflows and change requests from customers for customized products.
Monitoring this metric provides insights into how well products are being managed and coordinated within an individual plant. It is also a leading indicator of potential systemic quality problems in a product design or need for greater coordination with channel partners and customers.
3. Overall Equipment Effectiveness (OEE)
Defines how effective manufacturing workflows are to the machine and production line level, OEE is invaluable in benchmarking how specific manufacturing processes are performing. To gain the greatest insight using this metric, multiply Availability, Performance and Quality to capture the effectiveness of a specific machine or production line.
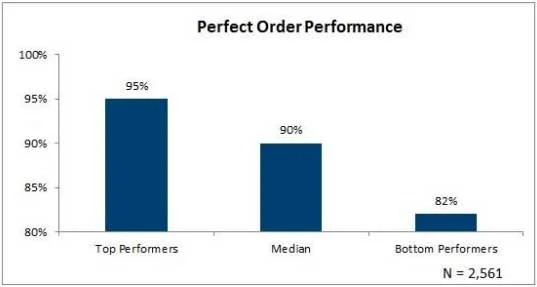
4. Perfect Order Performance
Provides an immediate measure of how well synchronized each step of order capture, order management, production and fulfillment are, perfect order performance is measured as an index value. To calculate the Perfect Order Index (POI), multiply % of orders delivered on time by % of orders completed by % of orders damage free by % of orders with accurate documents by 100.
The following is the equation broken out: (Percent of orders delivered on time) * (Percent of orders complete) * (Percent of orders damage free) * (Percent of orders with accurate documentation) * 100. According to e American Productivity and Quality Center (APQC), top performers have perfect order index scores between 90 and 95%.
5. Return Material Authorizations (RMAs) And Returns
RMAs are a direct measure of product quality and a products’ nonconformance to customers’ specifications and requirements. They are issued for a wide variety of reasons, and it’s important to have a monthly Pareto Analysis done of the top 20% of factors that drive 80% of the returns. This will make cause troubleshooting more efficient, leading to permanent solutions to problems that may be causing RMAs to begin with.
Sources:
Amer, Y., Luong, L., Lee, S. H., & Ashraf, M. A. (2008). Optimizing order fulfillment using design for six sigma and fuzzy logic. International Journal of Management Science and Engineering Management, 3(2), 83-99.
Motschman, T. L., & Moore, S. B. (1999). Corrective and preventive action. Transfusion Science, 21(2), 163-178.
Novack, R. A., & Thomas, D. J. (2004). The challenges of implementing the perfect order concept. Transportation Journal, 5-16.
